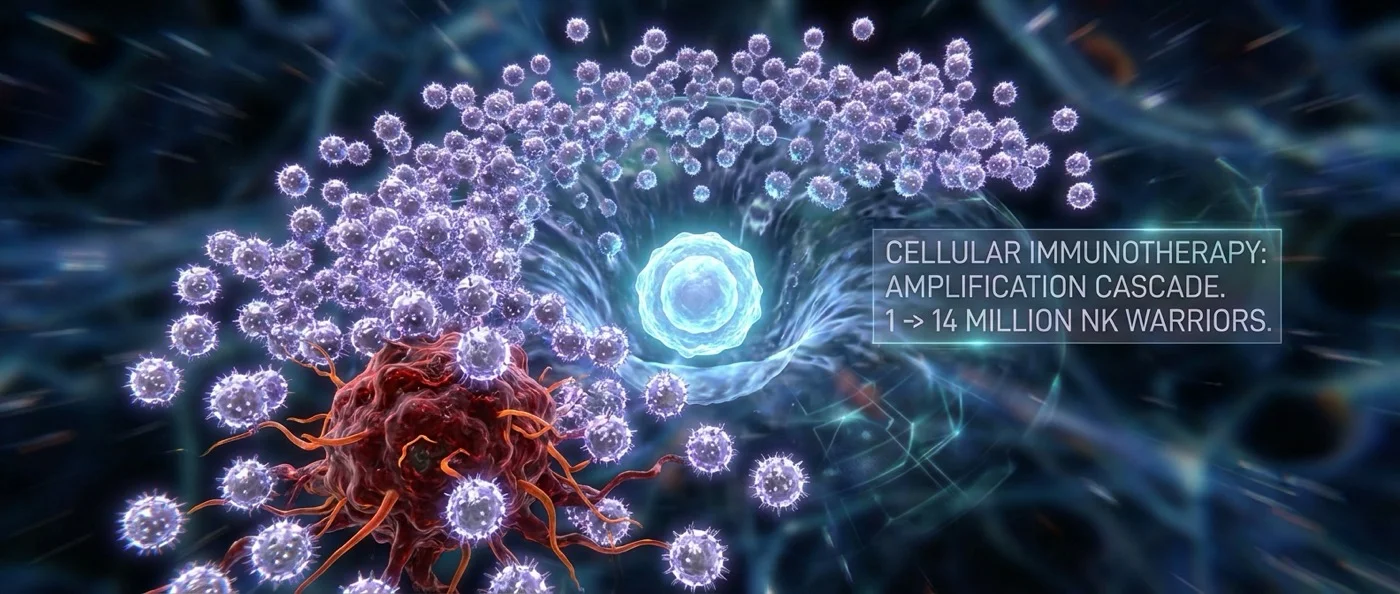
What if a single stem cell could produce 14 million warriors capable of destroying cancer cells? Researchers in China have just achieved exactly that — and their discovery promises to fundamentally reshape the landscape of cancer immunotherapy, making it cheaper, more effective, and more accessible.
🛡️ NK Cells: The Immune System's “Natural Born Killers”
Natural Killer (NK) cells are a unique class of white blood cells. Unlike T cells, which need to be “trained” to recognize an invader, NK cells automatically detect and destroy infected or cancerous cells — without prior exposure.
This ability makes them ideal tools for immunotherapy. In CAR-NK therapy, scientists equip NK cells with a lab-designed receptor (Chimeric Antigen Receptor) so they can recognize specific markers on cancer cells and attack them with precision. However, the traditional approach faced serious obstacles: high variability between cells, low genetic modification efficiency, and steep production costs.
🧬 The Innovation: Start From the Beginning
The team led by Professor Wang Jinyong at the Institute of Zoology of the Chinese Academy of Sciences chose a radically different strategy. Instead of modifying mature NK cells, they started with CD34+ hematopoietic stem and progenitor cells (HSPCs) isolated from cord blood. The logic was simple yet groundbreaking: by moving the genetic modification step earlier in the developmental chain, you gain efficiency at every stage.
🔬 Three Stages — One Stem Cell
The method involves three distinct stages that transform a single stem cell into millions of cancer fighters:
Expansion (14 days)
CD34+ HSPCs (or those already CAR-transduced) are cultured with irradiated AFT024 feeder cells. In just 14 days, they multiply 800 to 1,000 fold.
NK Commitment (Organoids)
Expanded cells are cultured with OP9 feeder cells, creating artificial hematopoietic organoid aggregates — structures that guide cells toward the NK lineage.
Maturation & Multiplication
Committed NK cells undergo full maturation and further expansion, producing highly pure iNK or CAR-iNK cells with endogenous CD16 expression.
The study, published in Nature Biomedical Engineering with Fangxiao Hu as first author, documents that a single CD34+ HSPC can generate up to 14 million iNK cells or 7.6 million CAR-iNK cells. The team estimates that one-fifth of a typical cord blood unit could theoretically yield thousands — perhaps tens of thousands — of therapeutic doses.
🎯 Results in Leukemia Models
The results were not just quantitatively impressive. In laboratory testing, both iNK and CAR-iNK cells demonstrated powerful anti-tumor activity. In mouse models of human B-cell acute lymphoblastic leukemia (B-ALL), CD19 CAR-iNK cells significantly reduced tumor growth rates and extended the animals' survival.
💡 Why This Discovery Matters
Current CAR-NK therapy requires massive amounts of viral vector for genetic modification of mature cells — one of the biggest cost barriers. The new method uses just 1/140,000 to 1/600,000 of that amount, dramatically cutting production costs. This means NK cell immunotherapy could soon become accessible to far more patients worldwide.
🔮 From Lab to Patient
The method doesn't just improve efficiency — it fundamentally changes the production philosophy. Instead of collecting NK cells from donors (with high biological variability), a cord blood bank could become the source of thousands of therapy doses — standardized, quality-controlled, and cheaper.
The concept of “off-the-shelf” immunotherapy that could be distributed like medication rather than custom-built for each patient now seems closer than ever. The research was funded by China's Ministry of Science and Technology and the National Natural Science Foundation, signaling the importance placed on this generation of immunotherapy.
Although clinical application still lies ahead, the laboratory data is remarkably encouraging: one source, one stem cell, millions of warriors. The fight against cancer just gained a powerful new weapon.