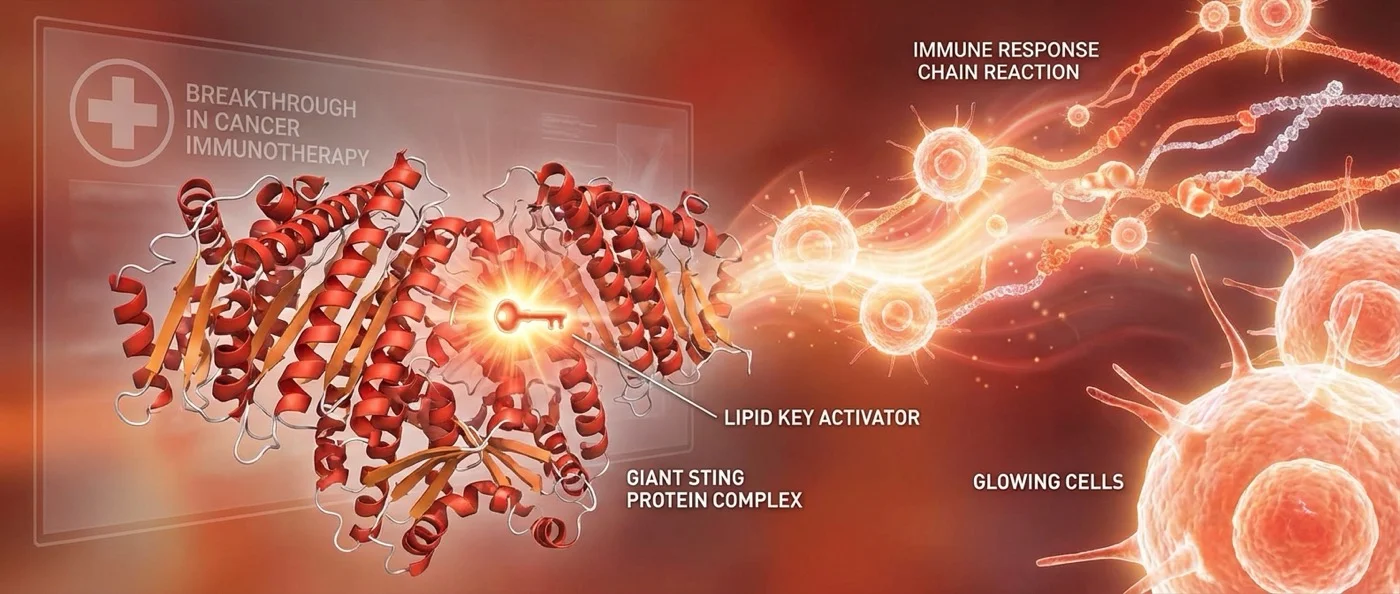
In the fight against cancer, the most promising strategy is no longer chemotherapy — it's activating our own immune system. At the heart of this revolution lies the STING mechanism — a molecular “alarm system” that detects threats inside cells. Recent research reveals that lipids play a key role in both activating and absorbing this mechanism — with enormous implications for treating cancer and autoimmune diseases.
🔔 What Is the STING Mechanism
STING (Stimulator of Interferon Genes) is a protein that functions as a “security system” inside cells. When a sensor protein — cGAS — detects DNA outside the nucleus (e.g. from a virus or damaged cells), it activates STING. STING positions itself on the cell membrane and triggers a chain reaction: it produces type I interferons, which summon T cells and Natural Killer (NK) cells — the immune system's “executioners.”
This mechanism is vital in both fields: in cancer, it can “awaken” the immune system against tumors. In autoimmune diseases, its overactivation can turn immune forces against healthy tissues.
🧬 The Hidden Power of Lipids
Researchers at Columbia University, led by immunologist Jordan Orange, made a striking discovery: Natural Killer (NK) immune cells protect themselves during attacks thanks to a dense “armoured” layer of lipids in their membrane. This “lipid shield” allows them to launch toxic molecules without suffering damage themselves.
NK Lipid Shield
NK cells have a denser lipid membrane than any other cell type — this protects them while they destroy cancer cells.
Cancer Copies the Shield
Certain aggressive cancer cells (e.g. breast) “copy” the lipid shield to evade immune attacks.
Weakening the Shield
Specific chemical compounds weakened the lipid membrane of cancer cells, making them vulnerable to NK attacks.
This discovery, published in PLOS Biology, opens an entirely new front: if you can weaken a tumor's “lipid shield” without affecting NK cells, you unlock an extremely targeted therapy.
"We don't know yet if this is a general mechanism by which cancer cells resist natural killer cells. If it is generalizable, we can start to think of therapies that disrupt the tumor cell membrane and make it more susceptible to attack by the immune system."
— Yu Li, Researcher, Columbia University💊 STING Agonists: A New Generation of Therapies
Directly activating STING through drugs — so-called “STING agonists” — is one of the most ambitious strategies in immunotherapy. However, the major challenge has always been precision: STING agonists activate the immune system throughout the body, causing excessive inflammation and serious side effects.
In September 2025, researchers at the University of Cambridge, led by Gonçalo Bernardes, published an elegant solution in Nature Chemistry: a two-part drug-delivery system where the STING agonist MSA2 is chemically “caged” and only released when it encounters an enzyme (β-glucuronidase) exclusively produced by tumors.
❌ Old Approach
STING agonists activate everywhere in the body → excessive inflammation, healthy tissue damage, serious side effects.
✅ New Cambridge Strategy
"Caged" drug activates only inside the tumor → targeted immune response, zero side effects.
"This is like sending two safe packages into the body that only unlock and combine when they meet the tumor's unique chemistry. The result is a strong immune-activating drug that appears only where it is needed."
— Gonçalo Bernardes, University of Cambridge⚖️ Dilemma: Cancer vs Autoimmune Disease
The STING mechanism sits at the crossroads of two opposing medical needs. In cancer, we want to activate it to the maximum — to rally the immune system against tumors. In autoimmune diseases (lupus, rheumatoid arthritis, psoriasis), STING overactivation has already been identified as a key mechanism: the body attacks itself.
This means that lipid-based regulation of STING could provide answers in both fields: controlled activation within tumors, or targeted inhibition in autoimmune tissues. Understanding which lipids activate and which inhibit becomes a critical factor.
💡 Nanotechnology + STING = A New Era
At Northwestern University, researchers redesigned the classic chemotherapy drug 5-fluorouracil (5-Fu) into spherical nucleic acids (SNAs) — nanostructures that embed the STING agonist in a DNA carrier. The result: 20,000 times more potent anti-cancer activity, 12.5 times better absorption by leukaemia cells, and zero side effects in animal models (ACS Nano, 2025).
🔮 The Future: Lipids as Therapeutic Targets
These discoveries open multiple therapeutic fronts:
- Targeted STING activation — “caged” agonists that unlock only inside tumors (Cambridge, Nature Chemistry)
- Weakening tumor lipid shields — compounds that make cancer cells vulnerable to NK attacks (Columbia, PLOS Biology)
- Lipid nanocarriers — spherical nucleic acids that deliver drugs with precision into cells (Northwestern, ACS Nano)
- STING inhibition in autoimmune diseases — regulating lipid composition to reduce inflammation in chronic conditions
- Combination therapies — STING agonists + checkpoint inhibitor immunotherapy for “cold” tumors
The convergence of lipid biology, immunology, and nanotechnology is creating a new field: targeted immunoreceptive therapy. Instead of bombarding the entire body, we send “smart packages” that unlock only where needed — whether that's a tumor or an inflamed tissue. For the first time, cancer may be fought by our own cells — as long as we give them the right signal.