The Bohr model explains atomic stability by introducing quantized electron orbits. How Niels Bohr incorporated quantum ideas into the atomic model.
📖 Read more: Marie Curie: How Radioactivity Opened the Nuclear Age
⚛️ The Problem of the Classical Atom
In the early 20th century, Ernest Rutherford proposed an atomic model where electrons orbit a positively charged nucleus — like a microscopic solar system. The picture was elegant, but it concealed a fatal flaw: according to Maxwell's classical electromagnetic theory, any charged particle that accelerates emits electromagnetic radiation.
An electron in a circular orbit is constantly accelerating (centripetal). This means it should continuously lose energy by emitting electromagnetic waves. As its energy decreases, its orbit would spiral inward until it crashes into the nucleus. Calculations showed this collapse would happen in <10⁻¹¹ seconds — less than one hundredth of a nanosecond.
According to Maxwell's equations, an electron orbiting a proton would lose all its energy in approximately 10⁻¹¹ seconds. Every atom in the Universe should have collapsed immediately after its creation. The existence of stable matter was classically impossible — something had to fundamentally change in our understanding.
Yet matter exists. Atoms are stable, electronic structures persist, and chemistry works. This contradiction between theory and reality was one of the greatest crises in early 20th-century physics — and the solution would come from a young Danish physicist.
💡 Bohr's Idea (1913)
Niels Bohr, born in Copenhagen in 1885, worked in Rutherford's laboratory in Manchester, where he confronted the problem of atomic stability face to face. In July 1913, he published a series of three papers titled «On the Constitution of Atoms and Molecules» that would change physics forever.
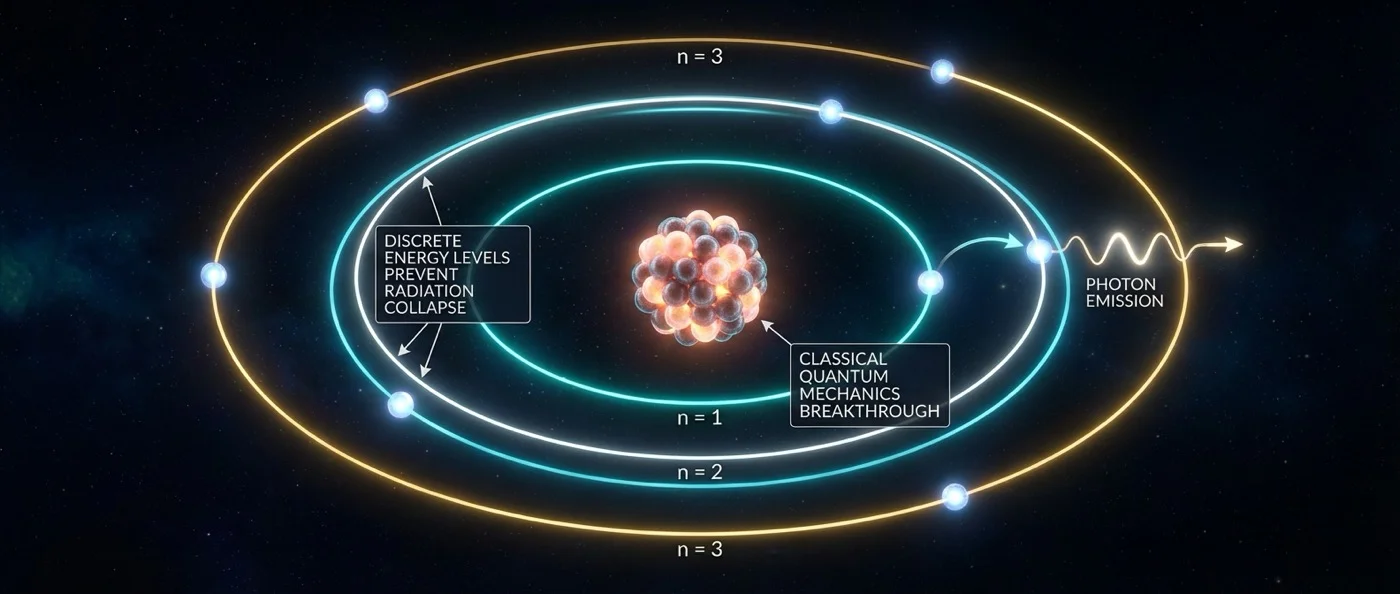
Bohr proposed three bold postulates. First, electrons can only move in specific orbits — so-called stationary states. Second, when in these orbits, electrons do not emit radiation, despite their continuous acceleration — an idea in direct contradiction with classical electrodynamics. Third, radiation is emitted or absorbed only when an electron jumps (quantum jump) from one allowed orbit to another.
Bohr's fundamental quantization condition stated that the electron's angular momentum must be an integer multiple of Planck's constant: L = nℏ, where n = 1, 2, 3, ... and ℏ = h/2π. This simple yet revolutionary idea meant that not all orbits are permitted — only those satisfying the quantization condition. Energy is not emitted continuously, but in discrete quanta.
Bohr thus incorporated Planck's constant — introduced in 1900 for black-body radiation — directly into the structure of the atom. The quantum idea no longer concerned only light, but matter itself.
🌈 Explaining the Hydrogen Spectrum
The greatest triumph of the Bohr model was explaining hydrogen's atomic spectrum. Since the late 19th century, physicists knew that hydrogen emits light at very specific wavelengths — discrete lines in the spectrum rather than a continuous distribution. Johann Balmer had already discovered in 1885 the empirical formula describing these lines in the visible spectrum (Balmer series), but no one could explain why these lines exist.
Bohr demonstrated that each spectral line corresponds to an electron transition between two energy levels. The energy of the emitted photon equals exactly the energy difference between the initial and final levels: E_photon = E_initial − E_final = hf, where f is the frequency of light. This formula perfectly reproduced the Balmer formula and the generalized Rydberg formula.
The Balmer series (transitions to n=2) corresponds to visible light. The Lyman series (to n=1) produces ultraviolet radiation, while the Paschen, Brackett, and Pfund series (to n=3, 4, 5 respectively) emit infrared. All these series follow the Bohr model's predictions exactly — a quantitative success that impressed the scientific community.
🔢 The Quantum Number n
In the Bohr model, each allowed orbit is characterized by the principal quantum number n (n = 1, 2, 3, ...). The energy of each level is given by the formula:
E_n = −13.6 / n² eV
The ground state corresponds to n=1, with energy −13.6 eV — this is hydrogen's ionization energy, the energy required to completely detach the electron from the nucleus. Excited states (n=2, 3, 4, ...) have higher energy (less negative) and larger orbital radii.
The orbital radius increases with n²: r_n = n² × 0.529 Å (Bohr radius). This means the first orbit (n=1) has a radius of 0.529 Å, the second (n=2) 2.116 Å, the third (n=3) 4.761 Å, and so on. Angular momentum is quantized as L = nℏ, meaning only integer multiples of the reduced Planck constant are allowed.
As n → ∞, the energy approaches zero and the radius tends to infinity — the electron “escapes” and the atom ionizes. Theoretically, infinite energy levels exist, but in practice very high ones (Rydberg atoms) are extremely sensitive to external perturbations.
🔬 Limitations of the Model
Despite its triumphant explanation of hydrogen's spectrum, the Bohr model had significant limitations. The first and most obvious was that it only worked for hydrogen-like atoms — atoms with a single electron (hydrogen, He⁺, Li²⁺). For multi-electron atoms, the model failed completely because it could not handle electron-electron interactions.
Second, the model could not explain the fine structure of spectral lines — the splitting of each line into closely spaced lines, which depends on relativistic corrections and spin-orbit coupling. Nor could it explain the Zeeman effect — the splitting of spectral lines in the presence of a magnetic field.
Third, the model treated the electron as a point particle in a defined orbit, ignoring its wave nature — something that would prove fundamental within the next decade. Bohr had taken a correct step, but understanding the full picture required radically new tools.
"If quantum mechanics hasn't profoundly shocked you, you haven't understood it yet." — Niels Bohr
🚀 From Bohr to Quantum Mechanics
The Bohr model, despite its limitations, paved the way for the full quantum theory. In 1924, Louis de Broglie proposed that electrons — and all particles in general — possess wave properties. The de Broglie wavelength is given by λ = h/p, where p is momentum. This idea immediately explained Bohr's quantization condition: the allowed orbits are precisely those in which an integer number of wavelengths fit — standing waves around the nucleus.
In 1926, Erwin Schrödinger formulated his own version of quantum mechanics based on the wave equation. The Schrödinger equation describes the electron not as a point particle in an orbit, but as a wave function ψ(r) distributed in space. |ψ|² gives the probability of finding the electron at a given position — the classical orbit was replaced by a probability cloud.
The transition from orbits to orbitals was a conceptual revolution. In modern quantum mechanics, an orbital is not a path but a three-dimensional region of space with a characteristic shape (s, p, d, f) and energy. The electron does not “travel” along a specific route — it exists as a probability wave until the moment of measurement.
Meanwhile, Werner Heisenberg developed matrix mechanics (1925), which proved mathematically equivalent to Schrödinger's wave mechanics. Heisenberg's uncertainty principle (Δx · Δp ≥ ℏ/2) sealed the transition: we can never simultaneously know both the position and momentum of an electron with precision — the very concept of an “orbit” loses meaning at the quantum level.
The Bohr model stands today as a historical bridge: the first successful synthesis of classical physics and quantum ideas, which led us to the framework of understanding we still use today. From Planck to Bohr, and from Bohr to Schrödinger, each step revealed that nature, at the smallest scale, does not operate by the rules of the “large world” — but by its own, deeper laws.