Scanning tunneling microscopes (STM) and atomic force microscopes (AFM) use quantum principles to “see” and manipulate individual atoms.
📖 Read more: DNA & Quantum Physics: How Tunneling Creates Mutations
💭 An Impossible Dream in Zürich
In the early 1980s, two researchers at the IBM laboratory in Zürich embarked on a project that many colleagues considered impossible. Gerd Binnig and Heinrich Rohrer wanted to build an instrument capable of “seeing” individual atoms — without lenses, without beams of light, without electron beams. Their idea was based on a phenomenon that quantum mechanics had predicted for decades but nobody had exploited for microscopy: quantum tunneling.
According to classical physics, an electron encountering an energy barrier cannot pass through it unless it has sufficient energy. Quantum mechanics, however, allows a small probability of “tunneling” — the electron can appear on the other side of the barrier even without the energy to overcome it. The tunneling current depends exponentially on distance: a change of just 1 Ångström (0.1 nm) is enough to alter the current by an order of magnitude.
🔬 The STM: First Contact with Atoms
Binnig and Rohrer exploited this extreme sensitivity. They fabricated an extraordinarily sharp metallic tip — so pointed that it ideally terminated in a single atom. They positioned the tip at a distance of 4 to 7 Ångströms (0.4–0.7 nm) from the surface of a sample and applied a voltage between them. Electrons “tunneled” through the vacuum, generating a current of a few tenths of a nanoampere. By scanning the tip across the surface — point by point, line by line — they recorded the current variations and converted them into an image.
The technique was named the Scanning Tunneling Microscope (STM). Their first publication came in 1982 in Physical Review Letters with the title “Surface Studies by Scanning Tunneling Microscopy.” But the image that captivated the entire scientific world came in January 1983: the 7×7 reconstruction of the silicon Si(111) surface, which corresponded to atomic resolution. For the first time, humanity “saw” individual atoms on a real surface.
"The confirmation that the 7×7 structure was real filled us with excitement — but also with the fear that nobody would believe us."
— Gerd Binnig, Nobel Lecture, 1986The doubt did not last long. The Nobel Committee awarded Binnig and Rohrer the 1986 Nobel Prize in Physics, shared with Ernst Ruska (inventor of the electron microscope). It was a rare recognition: the invention was honored just five years after its construction.
⚡ From Tunneling to Force: The Birth of the AFM
The STM had a fundamental limitation: it worked only on conductive or semiconducting samples because it required a tunneling current. Biological molecules, ceramics, polymers — vast categories of materials remained invisible. Binnig, together with Calvin Quate of Stanford and Christoph Gerber of IBM, found the solution: instead of measuring current, they would measure force.
The result was published in 1986 in Physical Review Letters: “Atomic Force Microscope.” The new microscope used a microscopic cantilever made of silicon or silicon nitride, with a sharp tip at its end. As the tip scanned across the surface, van der Waals forces, electrostatic forces, and atomic forces caused the cantilever to bend. A laser beam, reflecting off the back of the cantilever onto a photodiode (the beam-deflection method), recorded every variation with picometer precision.
The first commercial AFM was released in 1989. The first “true” atomic resolution under ambient conditions was achieved by Ohnesorge and Binnig in 1993. Franz Giessibl achieved atomic resolution of the Si(111) 7×7 surface using AFM shortly after, while modern AFMs have achieved subatomic resolution — imaging the electron density within a single atom.
🎨 The Night IBM “Painted” Atoms
One night in September 1989, researcher Don Eigler at IBM's Almaden Research Center in California used an STM at liquid helium temperature (4 K) to drag 35 xenon atoms across a nickel surface, spelling out the letters “IBM.” It was the first time a human had placed individual atoms in a pattern of their choosing — a milestone in nanotechnology. The image appeared on the cover of Nature and became one of the most recognizable symbols of science in the 20th century.
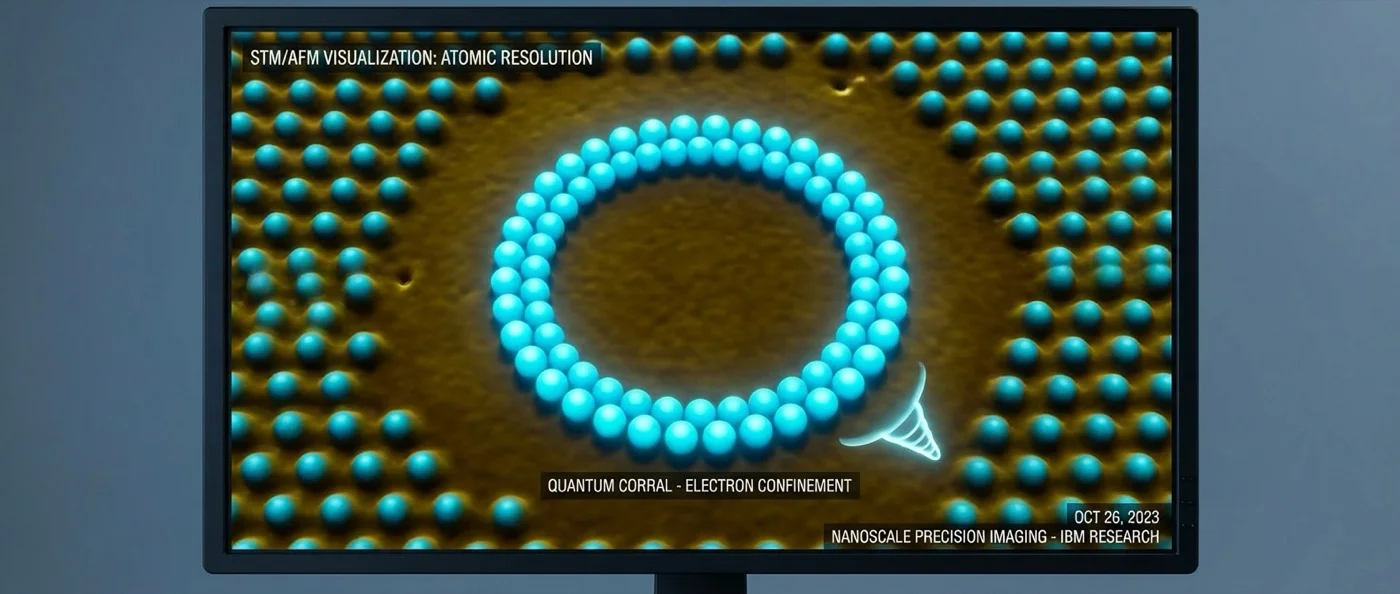
The same technique — atomic manipulation with STM — later enabled the creation of "quantum corrals": circular structures of individual iron atoms on copper, inside which electron wavefunctions formed visible patterns of standing waves (Friedel oscillations). Quantum mechanics was no longer abstract theory — it was literally visible in an image.
🌐 Beyond Atoms: Modern Applications
Today, the scanning probe microscopy (SPM) family includes dozens of variants. MFM (Magnetic Force Microscopy) maps magnetic fields at the nanoscale. KPFM (Kelvin Probe Force Microscopy) measures surface potentials. NSOM (Near-field Scanning Optical Microscope) surpasses the classical diffraction limit. The resolution of these microscopes is not limited by light diffraction — it is limited only by the probe-sample interaction volume, which can reach a few picometers, smaller than the diameter of a hydrogen atom.
In biology, AFMs study protein structures, cell mechanical properties (distinguishing cancerous from normal cells), and microtubules. In nanoelectronics, scanning probe lithography is used to create circuits at the atomic scale. In pharmaceuticals, AFM measures the binding forces between target molecules and drug compounds with picoNewton precision.
🏆 The Legacy of a Quantum Idea
From a needle and a quantum phenomenon in Zürich, scanning probe microscopy transformed the invisible world of atoms into a visible landscape. The discovery was not merely technological — it was philosophical. For centuries, atoms were theoretical entities, hypotheses of physicists and chemists. After the STM and the AFM, we could not only “see” them but also move them one by one.
Forty years later, quantum microscopy remains the backbone of nanotechnology — from semiconductor chips to biological vaccines. And at the heart of the technology lies the same principle: electrons can pass where classical physics says they cannot.