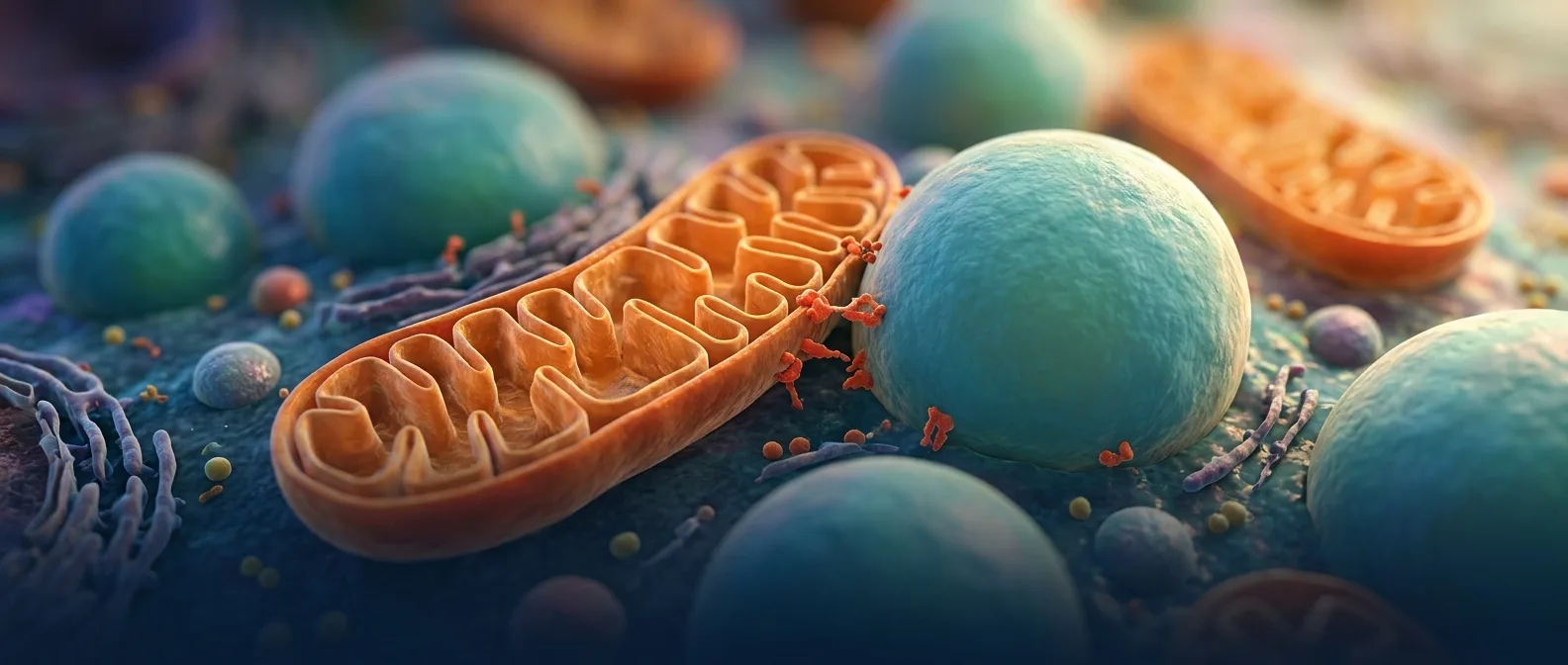
Less than a year ago, in a university cell biology lab, a microscope captured something stunning. Mitochondria and lipid droplets — two cellular structures thought to be relatively independent — appeared locked in an intricate dance. This "partnership" discovered in 2026 rewrites what we know about liver metabolism and the mechanisms behind fatty liver disease.
Researchers from Nature Metabolism revealed that the protein perilipin 5 (PLIN5) acts as a molecular switch, controlling exactly how mitochondria interact with lipid droplets in hepatocytes. PLIN5 phosphorylation — essentially adding a phosphate group — determines whether these two structures will "cooperate" effectively or not.📖 Read more: Mitochondria: Ancient Aliens Living Inside Every Cell
🔬 How Mitochondria-Lipid Droplet Contact Works
Picture mitochondria as energy factories that need constant fuel delivery. Lipid droplets are the warehouses storing that fuel — fatty acids that can burn for energy. But how does the "fuel" transfer efficiently from warehouse to factory?PLIN5 functions as a molecular "bridge" connecting these two structures. When properly phosphorylated, the protein makes mitochondria stick to lipid droplets, creating a direct lipid transfer channel.
This process isn't random. Research showed that during fasting, mitochondria-lipid droplet contacts increase dramatically, while in animals fed a Western diet (high in fats), these contacts are rare. What does this mean for the organism?Adaptation to Nutrient Availability
During fasting, the liver receives increased fatty acid flow from adipose tissue. Hepatocytes must manage this lipid "tsunami" without being destroyed by lipotoxicity. Increased PLIN5 and more mitochondrial contacts help direct lipids toward oxidation for energy production. Conversely, in abundance (like Western diet), this adaptive mechanism appears disrupted. While this might initially sound paradoxical, the explanation lies in system overload.📖 Read more: Microbiome: 38 Trillion Bacteria That Control Your Body
⚡ Phosphorylation: The Molecular Switch
The research focused on specific PLIN5 phosphorylation sites, particularly serine 155. Researchers created two variants:S155A Mimics non-phosphorylated state
S155E Mimics permanently phosphorylated state
Protection from Lipotoxicity
Particularly significant: S155A variant overexpression in Western diet-fed animals reduced lipotoxicity. This suggests proper mitochondrial contact management can protect hepatocytes from damage. But why is this protection so critical? When fatty acids accumulate freely in the cytoplasm, they can cause oxidative stress and inflammation — two key factors driving fatty liver disease.📖 Read more: Ocean Currents: How Invisible Rivers Control Our Planet
🧬 Spatial Distribution and Zonal Organization
One of the research's most intriguing findings concerns liver spatial organization. Hepatocytes aren't all identical — there's clear zonation from periportal to pericentral regions. In periportal areas, mitochondria had higher sphericity and lower density. Conversely, in pericentral regions, morphology was more tubular with increased density. Lipid droplets were sparse in periportal areas but increased dramatically toward pericentral zones.This zonal organization reflects the liver's functional dichotomy — lipid oxidation versus synthesis and storage.
Nature Metabolism, 2026
New Methodology: scPhenomics
Researchers developed an innovative technique called "scPhenomics" — a single-cell phenotypic analysis method. This approach enabled mapping lipid handling across different nutritional states with unprecedented resolution. Using confocal microscopy images from transgenic mice expressing mitochondrial Dendra2, the team could track organelle interactions in real-time.📖 Read more: 12-Meter Crystal Cave: Life Thrives in Extreme Heat
📊 Clinical Implications for MASLD
Metabolic dysfunction-associated steatotic liver disease (MASLD), affecting roughly one-third of adults globally, represents a serious health challenge. This research's findings open new therapeutic intervention pathways. Targeting signaling pathways that regulate PLIN5 phosphorylation could constitute a new strategy for preventing or reversing steatosis. Specifically, activating AMPK (AMP-activated protein kinase), responsible for PLIN5 phosphorylation, could improve mitochondrial-lipid coupling.Pharmaceutical Interventions
Developing small molecules that modulate PLIN5 phosphorylation
Diagnostic Biomarkers
Using phosphorylation status as early liver dysfunction indicator
Gene Therapy
Targeted expression of specific PLIN5 variants